What Single-Dose Gene and Cell Therapies Actually Do
Single dose gene therapy and related cell-based treatments aim to fix disease at its source with a one time infusion treatment, rather than repeated pills, injections or infusions. Traditional therapies usually manage symptoms by continuously replacing something the body lacks or blocking harmful pathways. Gene and cell therapies instead try to reprogram the body’s own cells so they produce the missing protein or stop causing damage. Often delivered through viral vectors or edited cells, they act like a biological software update: once the new code is installed, it keeps running. That promise of a “functional cure” is powerful, but it also raises questions. How long will the effect last? Can it be safely repeated if it fades? And who will be able to access such complex, highly specialized treatments as they move from rare disorders to more common diseases?
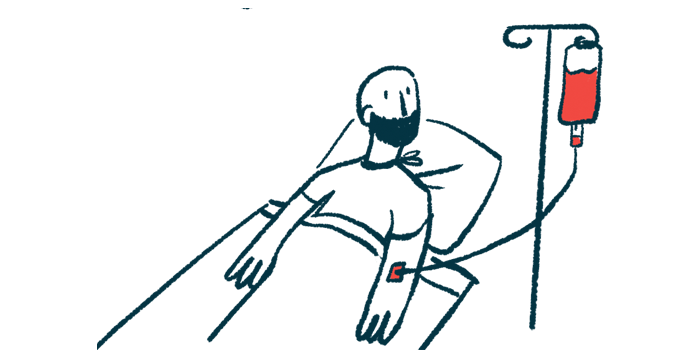
Sickle Cell Gene Therapy: Turning Off the Sickle Switch
Sickle cell gene therapy offers a vivid example of this shift. At UCLA Health, a young man named Samuel Idahosa grew up with undiagnosed sickle cell disease, enduring frequent hospitalizations and strict limits on physical activity. Sickle cell disease deforms red blood cells into rigid, crescent shapes that clog blood vessels, trigger excruciating pain, and shorten cell lifespan, leading to chronic anemia. In a leading-edge treatment, physicians harvested his cells and used genetic tools so his red blood cells would no longer sickle. After this single, intensive intervention, his clinicians now consider him cured, and his daily life has been transformed. Instead of relying on lifelong medications and hospital visits, his blood system has been fundamentally reprogrammed. This sickle cell gene therapy illustrates how one well-targeted procedure can replace years of conventional care—while also highlighting the need to track long-term safety and durability.
Roctavian for Hemophilia A: A One-Time Infusion That Lasts Years
The approved gene therapy Roctavian for severe hemophilia A is another landmark in one time infusion treatment. People with hemophilia A lack sufficient factor VIII, a clotting protein, and traditionally depend on frequent factor VIII replacement therapy to prevent bleeding episodes. Roctavian uses a modified adeno-associated virus to deliver a working factor VIII gene to liver cells, enabling the body to make its own clotting factor. In a Phase 1/2 clinical trial, a single infusion of Roctavian led to sustained reductions in bleeding and prophylactic treatment use for up to seven years. Dose mattered: participants receiving 6×10¹³ vector genomes per kilogram experienced more robust and durable factor VIII activity than those given 4×10¹³. Importantly, researchers reported no concerning long-term safety signals in this study. Roctavian hemophilia A data show how dose, durability and safety are tightly linked in single dose gene therapy, and why long-term follow-up is crucial.
Single-Experience Neuroplasticity: Rewiring the Brain in One Shot
Single-shot interventions are not limited to blood disorders. In neuroscience, researchers are uncovering forms of single experience neuroplasticity that can rewire brain circuits after just one event. Work in the hippocampus, a brain region crucial for memory, shows that dendrites—the branching structures of neurons—generate complex electrical events called plateau potentials. These long-lasting surges can prime a neuron to fire more easily and reshape its connections, effectively encoding a new memory in a single pass through an environment. Studies of behavioral timescale synaptic plasticity suggest that even individual neurons can perform deep, network-level computations, thanks to this dendritic activity. While these findings are still in the laboratory, they hint at future therapies that might harness one-time brain stimulation or precisely timed experiences to permanently adjust pathological circuits, from chronic pain to mood disorders, blurring the line between a procedure and a lasting cure.
Risks, Unknowns and a Near-Future of One-Time Interventions
Despite their promise, single-dose therapies bring new uncertainties. Because they act for years, unexpected side effects may emerge long after treatment, and repeating the same gene vector might be difficult if immunity develops. Some patients could still need retreatment as benefits wane, challenging the idea of a lifetime cure. Access and ethics loom large: who gets priority when therapies promise functional cures but require specialized centers and complex manufacturing? As more conditions, from rare genetic disorders to common diseases, move toward one-time interventions, insurers and health systems will need new models that weigh high upfront effort against long-term reductions in hospitalizations and chronic care. Parallel advances—like single-cell atlases that map how conditions such as Down syndrome reshape brain development, or platforms that predict cancer risk from how individual cells respond to stress—suggest a future where deeply personalized, sometimes one-off interventions become central to medicine.

