Most of the Universe’s Water Is Frozen in Space
When you paddle out beyond the break or dive beneath a wave, it is easy to think of oceans as the natural home of water. Yet most of the universe’s water never becomes liquid at all. In deep interstellar clouds, temperatures can fall to around 10 Kelvin, so low that water freezes as a thin coating on dust grains. These tiny grains act like cosmic chemistry labs, building water molecule by molecule in conditions far harsher than anything on Earth. Astronomers now know that water in space often forms long before planets, seas, or even complete star systems exist. That means the liquid rolling toward the shore today has a history stretching back to these cold, dark nurseries. Understanding water in space helps scientists piece together the origin of Earth oceans and reveals how extraordinary it is to have a world where water can flow freely in rivers, lakes, and waves.

Interstellar Comet Water: 3I/ATLAS and Its Semi-Heavy Ice
One of the most intriguing clues about water in space comes from the interstellar comet 3I/ATLAS, a rare visitor passing through our solar system. Observations with the Atacama Large Millimeter Array showed that this comet’s ice contains an astonishing amount of semi-heavy water, where one hydrogen atom is replaced by deuterium. Semi-heavy water is uncommon in nature, and previous measurements of comets revealed they typically hold more deuterated water than Earth’s oceans, suggesting they are not the main source of our seas. 3I/ATLAS is even more extreme: its ratio of semi-heavy to normal water is about 30 times higher than comets born in our own system and more than 40 times Earth’s oceans. To create that much deuterated water, the comet must have formed in an incredibly cold environment, far below minus 200 degrees Celsius, under very specific radiation conditions in a foreign planetary system.

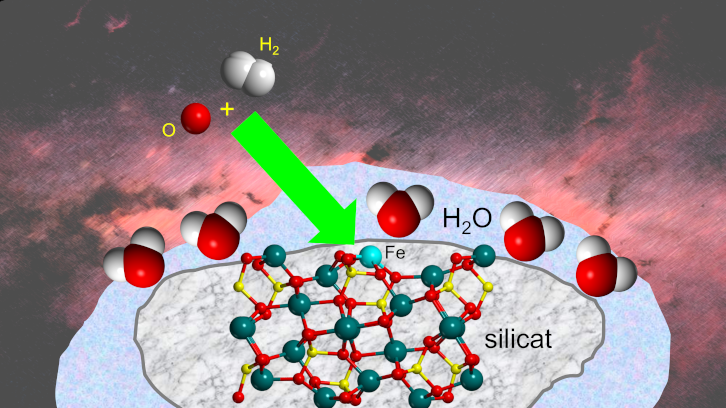
Cosmic Grains as Tiny Factories for Water in Space
Beyond spectacular comets, much of the universe’s water forms quietly on microscopic dust grains drifting between stars. Recent simulations highlight how iron-rich silicate grains, such as those containing the mineral olivine, can act as efficient water factories. In the frigid vacuum of space, simple gas reactions are too slow to explain the abundance of water observed. Instead, dust surfaces provide a place where atoms and molecules can meet. Iron ions embedded in these grains can grab onto hydrogen molecules and weaken their bonds, priming them for reaction. When an oxygen atom lands nearby, its arrival releases energy that helps split the hydrogen molecule apart. One hydrogen sticks to the iron, the other to the surrounding silica, ultimately combining with oxygen to form water. Over millions of years, this process coats grains with icy mantles. These icy particles, once part of interstellar clouds, may later become building blocks of planets and comets.
From Cosmic Ice to the Origin of Earth Oceans
All this distant chemistry feeds into a central question: what is the origin of Earth oceans? Theories suggest that early Earth, born hot and hostile, may have received at least part of its water from icy bodies and dust leftover from star formation. As comets and water-rich asteroids bombarded the young planet, they could have delivered frozen water made long before in interstellar clouds. However, measurements of semi-heavy water in many comets show higher deuterium levels than in our seas, including the extreme case of interstellar comet water from 3I/ATLAS. That mismatch implies comets alone cannot explain the exact composition of Earth’s oceans. Instead, a mix of sources is likely: water bound in minerals of the forming planet, icy dust grains, and some cometary contributions. The story of cosmic ice and oceans is therefore a complex blend, but it underlines a key point: our liquid oceans are the product of rare, finely tuned cosmic history.
Why Surfers, Swimmers, and Paddlers Should Care About Water in Space
For anyone who loves surfing, open-water swimming, or paddling at sunrise, space science might seem distant. Yet learning about water in space deepens appreciation for how unusual Earth’s oceans are. Interstellar comets like 3I/ATLAS show that water can exist in many forms and chemistries, often locked as ice at unimaginably low temperatures. Cosmic dust grains reveal how hard the universe has to work just to assemble a single water molecule. Against that backdrop, a planet covered in liquid seas, warm enough for waves and marine life, is an extraordinary exception. Knowing how rare a stable, accessible water environment may be turns every session on the water into something precious. It also adds weight to ocean conservation: pollution, overuse, and warming threaten a system that took cosmic timescales to assemble. Looking up at the night sky can remind us that this blue playground is both fragile and irreplaceable.
