Left, Right, and the Mystery of Life’s Single Hand
Many of life’s essential molecules come in chiral forms: mirror-image shapes that cannot be superimposed, like left and right hands. Amino acids can be left- or right-handed, and sugars that build genetic molecules also have two mirror versions. Yet living cells overwhelmingly prefer just one hand of each. Proteins are built almost entirely from left-handed amino acids, while the sugars in DNA and RNA are right-handed. This consistent choice is called homochirality, and it underpins the precise lock-and-key fit of enzymes, receptors, and genetic structures. Mix both hands randomly and those locks jam; biological machinery becomes unreliable. The puzzle is why life uses one hand instead of an even mix that ordinary chemistry tends to produce. Solving this means asking how tiny physical biases, hidden inside individual molecules, could have nudged early chemistry toward one side and then locked that choice in.

Electron Spin Biology: How Moving Charges Break Molecular Symmetry
Recent work on chiral molecules suggests that electron motion and spin can subtly favor one molecular hand over the other. Electrons carry a quantum property called spin, which behaves a bit like a tiny compass needle pointing in particular directions. When electrons move through chiral molecules, their spin and path become linked, a phenomenon known as chirality-induced spin selectivity. Experiments with gold and silver films coated in chiral layers, as well as short chains of amino-acid-like units, revealed unequal spin-linked electrical signals between left-handed and right-handed forms. Energetically, the two mirror images remain identical at rest, so the imbalance only appears when electrons flow, react, or interact with magnetized surfaces. That matters because real chemistry, especially in living systems, is full of moving charges and collisions. Even a modest spin-driven preference could, over countless reactions, tilt prebiotic chemistry toward one consistent molecular handedness.

Homochirality Explained: From Quantum Quirks to Working Drugs
This electron-spin bias adds a physics-based layer to older ideas about why life is homochiral. Previous explanations emphasized chance imbalances, mineral templates, or radiation that might slightly favor one hand of a molecule. Spin-driven effects offer a mechanism that acts during actual reactions, not just in static crystals. Over time, such biases could steer molecules like ribo-aminooxazoline toward one form when they crystallize on magnetic minerals, letting that hand dominate. The practical stakes are huge: many modern drugs are chiral, and the wrong hand can be ineffective or harmful because it fits a biological target differently. Understanding how electron spin interacts with molecular handedness could help chemists design more selective reactions, smarter pharmaceuticals, and new materials that exploit spin-filtering. It also reframes electron spin biology as more than an abstract quantum curiosity; it becomes a hidden lever that helped set life’s basic architecture.
One-Sided Planets and Other Asymmetries in Nature
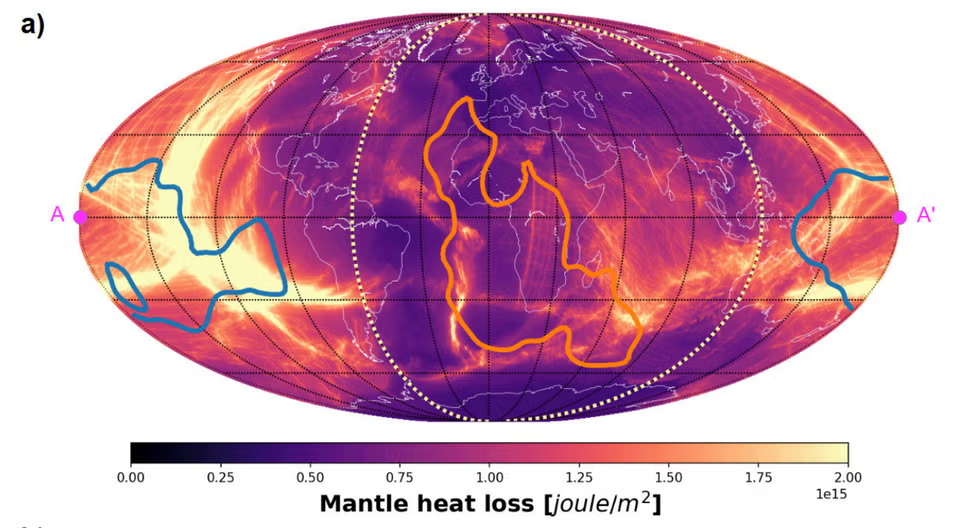
Life’s single-handed chemistry is not the only example of nature favoring one side. Deep inside Earth, heat from the molten interior leaks out unevenly. Models of the past hundreds of millions of years show that the hemisphere beneath the vast Pacific has cooled faster than the hemisphere beneath the large landmasses of Africa, Europe, and Asia. Thin oceanic crust and enormous volumes of cold seawater let interior heat escape more efficiently than thick continental regions, which act like insulation. Over geological time, this has created a planetary-scale thermal imbalance: one side effectively sheds heat quicker. Just as a small molecular twist can bias electron flow, a difference in crust thickness and ocean coverage can bias planetary cooling. Both cases show how symmetry in initial conditions does not guarantee symmetric outcomes. Tiny or local asymmetries can accumulate, shaping everything from tectonic patterns to the chemistry of emerging life.
How Scientists Probe Electron Spin and Chiral Molecules
To study electron spin and molecular handedness, researchers combine precise materials with sensitive electrical measurements and simulations. In the lab, they deposit thin metallic films, such as gold or silver, and coat them with chiral molecules or build chiral layers directly into the material. By driving a small electrical current through these structures and monitoring how the signal changes with spin orientation or magnetic alignment, they can detect tiny differences between left- and right-handed forms. These experiments are backed by ab initio calculations, which track electron behavior using fundamental physics rather than adjustable parameters. Simulations show that, even when mirror-image molecules share identical energies, their internal spin orientations differ. Together, these approaches reveal how moving electrons experience a chiral landscape differently depending on their spin. The result is a clearer, experimentally grounded picture of how quantum-scale effects can influence chemical reactions and, ultimately, biological structure.