Seeing Cells One by One: The New Resolution of Biology
For most of modern medicine, cells were studied in bulk, like tasting a blended smoothie instead of the individual fruits. Single cell technology changes that. By reading out genes, proteins and signaling pathways cell by cell, scientists can see how even “identical” cells behave differently, respond to drugs, or turn cancerous. New optical nanoscopy imaging methods push this still further, following events at the level of single molecules and ions in real time. Together, these tools turn living tissue into a dynamic map rather than a static snapshot. For patients, that resolution matters. It helps explain why one person’s tumour melts away on immunotherapy while another’s barely shrinks, or why a gene therapy works beautifully in some tissues but not others. This emerging view of biology as a shifting mosaic of distinct cells underpins the future of precision medicine.
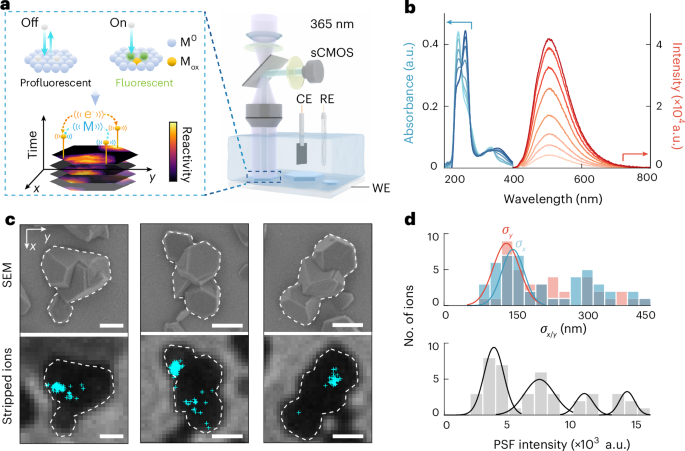
Optical Nanoscopy at Single-Ion Scale: Tiny Signals, Big Medical Implications
A recent advance in optical nanoscopy imaging has made it possible to track individual ions as they move in and out of tiny metal structures during “stripping” reactions. Originally developed to understand how battery materials charge, discharge and eventually fail, this technique can follow spatiotemporal ion dynamics with single-ion and subparticle resolution. That kind of control is highly relevant to future bioelectronic and implantable devices, which depend on precise ion flows at material–tissue interfaces. Being able to watch ions in action lets engineers design surfaces that are more stable, less prone to degradation and friendlier to surrounding cells. In turn, this could lead to smarter neural implants, long-lived biosensors and finely tuned drug delivery systems that respond to local chemistry in the body. The same physics guiding better batteries is now informing how we might power and protect next-generation medical hardware.
SEPARATE‑Seq and Tumor Immune Mapping: Rewriting the Map of Lung Cancer
In lung adenocarcinoma, the most common form of lung cancer, the difference between immune cells that truly infiltrate a tumour and those just passing in nearby blood can decide whether immunotherapy works. Researchers at VIB and Vrije Universiteit Brussel tackled this with a new approach, combining a patient-relevant mouse model with advanced single cell technology, often described under the banner of SEPARATE‑Seq lung tumour immune mapping. By letting tumours grow directly in the lung and then separating tumour nodules from nearby healthy tissue, they built one of the most detailed immune maps of this cancer type to date. The model recapitulates key immune features seen in patients, such as dysfunctional natural killer cells and increased regulatory and exhausted T cells. This refined tumour immune mapping offers a clearer roadmap for designing or sequencing immunotherapies so they can reawaken the right immune cells in the right place.
Connecting the Dots: From In‑Body Gene Editing to Cell‑Level Targeting
The first demonstrations of in body gene editing showed that CRISPR-based systems could be delivered directly into a person to correct disease-causing DNA changes inside their tissues. That leap opened the door to treating genetic conditions at their source, but it also raised questions: which cells should be edited, how many, and with what long-term consequences? This is where single cell technology and tumour immune mapping become essential. By charting the exact cellular neighborhoods inside organs or cancers, researchers can identify which cell types drive disease and which should be left untouched. Optical nanoscopy imaging, meanwhile, helps ensure that delivery vehicles, implants and bioelectronic devices behave predictably at microscopic interfaces. Together, these tools allow in body gene editing and other advanced therapies to move from blunt, organ-level interventions to finely targeted, cell-by-cell strategies that are both more effective and potentially safer.
What This Means for Patients Over the Next Decade
As these technologies mature, their impact will increasingly be felt in clinics. For cancer, detailed tumour immune mapping should enable oncologists to classify tumours not only by size and location, but by their cellular ecosystems, guiding which immunotherapy combinations are most likely to work. In genetic diseases, in body gene editing may be paired with single cell readouts to verify exactly which cell types have been corrected and to monitor off-target effects over time. Meanwhile, bioelectronic and implantable devices improved through optical nanoscopy insights could last longer, integrate better with tissue and deliver drugs or electrical signals with greater precision. Taken together, these advances point toward a future of precision medicine where diagnosis and treatment are tuned to each patient’s unique cellular landscape, turning once-unpredictable therapies into more reliable, personalised care.