Why Cancer Care Is Shifting Toward Finite, Smarter Treatment
For decades, cancer treatment has meant long cycles of chemotherapy, frequent hospital visits, and difficult side effects that can stretch on for months or years. Today, next generation oncology is moving toward something more precise: finite courses, single dose cancer therapy ideas, and tightly targeted combinations. The aim is not just to shrink tumors, but to do it quickly and deeply enough that patients can stop treatment earlier and live more normally. Two examples illustrate this shift. In myeloid leukemia of Down syndrome, researchers are using single cell sequencing to map how the disease evolves step by step, opening the door to highly tailored, possibly one-time interventions. In metastatic pancreatic ductal adenocarcinoma, an experimental RAS inhibitor called daraxonrasib is being tested in the first line pancreatic cancer setting alongside chemotherapy, showing striking early responses but also highlighting the trade-offs in toxicity. Together, these advances hint at a future where treatment is shorter, smarter, and more individualized.
Reading Cancer’s Playbook Cell by Cell
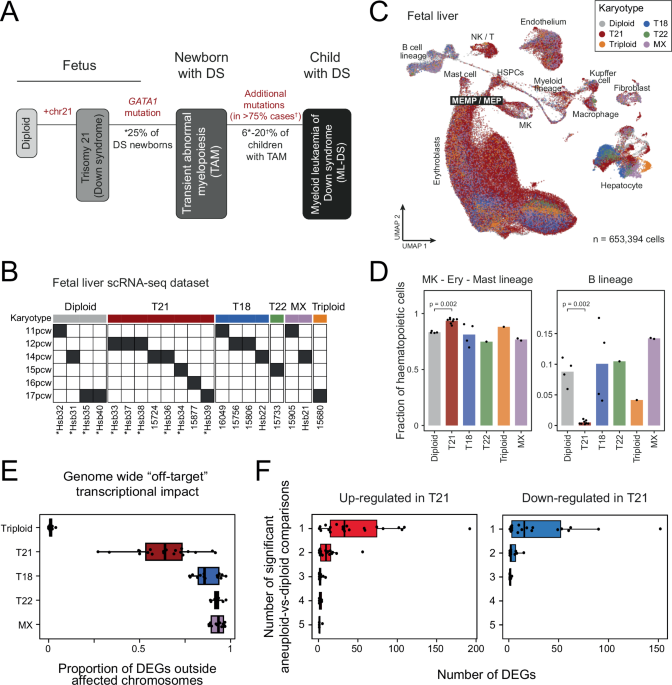
Myeloid leukemia of Down syndrome (ML-DS) is an aggressive blood cancer that often strikes young children, and standard chemotherapy can be especially harsh for them. Scientists are now dissecting this disease at single-cell resolution, using single cell RNA sequencing to capture the gene activity of thousands of individual cells from fetal liver and leukemia samples. By combining these data with whole genome sequencing and advanced computational tools, they reconstruct how normal blood cells gradually acquire mutations and shift into pre-leukemic and then fully leukemic states. This single cell transcriptional evolution acts like a time-lapse video of cancer forming. In the future, such detailed maps could pinpoint the narrow window when a single, highly targeted cancer treatment might erase dangerous cells before they expand, or guide short, intensive regimens tailored to the exact mutations and cell types driving a person’s disease. It is a foundational step toward truly precision, and possibly limited-duration, leukemia therapy.
Daraxonrasib in Pancreatic Cancer: Powerful First-Line Punch, Tough Side Effects
Pancreatic ductal adenocarcinoma has long been one of the toughest cancers to treat, with modest response rates to standard multiagent chemotherapy. A new RAS inhibitor, daraxonrasib, is being studied as a targeted cancer treatment in metastatic disease. In a phase 1/2 trial, 39 patients with RAS-mutant metastatic pancreatic cancer received daily oral daraxonrasib plus standard chemotherapy as first-line therapy. The objective response rate reached 59%, and the disease control rate was 92% at a median follow-up of 10 months, compared with historical response rates of around 30%–40% for chemotherapy alone. Earlier, a separate phase 3 study in previously treated patients showed that daraxonrasib monotherapy doubled overall survival relative to standard chemotherapy. These numbers suggest a major advance, but there is a cost: grade 3 or higher adverse events were common, and individual experiences, such as severe skin reactions, underline the toxicity risk. The challenge now is balancing this impressive efficacy with tolerability and quality of life.
From Continuous Chemo to Single Shots and Finite Combos
What ties these breakthroughs together is a broader move away from endless chemotherapy toward more defined treatment courses. In pancreatic cancer, daraxonrasib plus chemotherapy represents a next generation oncology approach: stacking a precision drug on top of standard drugs to hit tumors harder in the first line, with the hope of longer control after treatment ends. In ML-DS, detailed single cell sequencing and phylogenetic analysis are revealing which cell populations appear early, which mutations drive progression, and which clones resist therapy. That knowledge could eventually justify a one-time, aggressive strategy aimed at eradicating high-risk cells before they expand. Finite-duration strategies also appeal to patients and families: fewer infusions, shorter disruption of daily life, and potentially more durable responses. The key is knowing who can safely benefit from a one-shot or short-course approach and who still needs longer, gentler regimens.
What Patients Should Watch for in the Next Decade
Over the next 5–10 years, patients and families are likely to hear more about single dose cancer therapy concepts, highly tailored combinations, and first line pancreatic cancer trials built around new targeted agents. Precision tools such as single cell sequencing will increasingly guide which treatments are offered, helping doctors match specific mutations and cell states to specific drugs. At the same time, practical issues will matter: how regulators judge novel, high-impact but high-toxicity regimens; how insurers handle expensive short-course treatments versus traditional chemotherapy; and how side effects are monitored and managed in real life. When discussing options with clinicians, patients may want to ask whether any targeted cancer treatment or finite-duration regimen is available for their cancer type, how it compares with standard care, and what is known about long-term outcomes. The overall direction is hopeful: more precision, shorter courses, and treatments designed around each person’s unique disease biology.