From Tumor Snapshots to Single-Cell Detail
Traditional pathology views a tumor as a single mass: a slice of tissue under the microscope, averaged test results, and broad labels such as “aggressive” or “slow-growing.” In reality, every tumor is a mosaic made up of many different kinds of cells, each behaving in its own way. Some cells divide rapidly, others hide from the immune system, and still others may already be resistant to treatment. Single-cell tumor profiling is a new way to study cancers that reads out biological information from thousands to millions of individual cells within one tumor. Instead of blending all the signals together, it keeps the fine-grained detail. Scientists can see which genes are active in each cell and how the DNA packaging—called chromatin—opens or closes to control those genes. This level of detail matters because the rare but dangerous cells that drive relapse or spread can easily be lost when data are averaged.
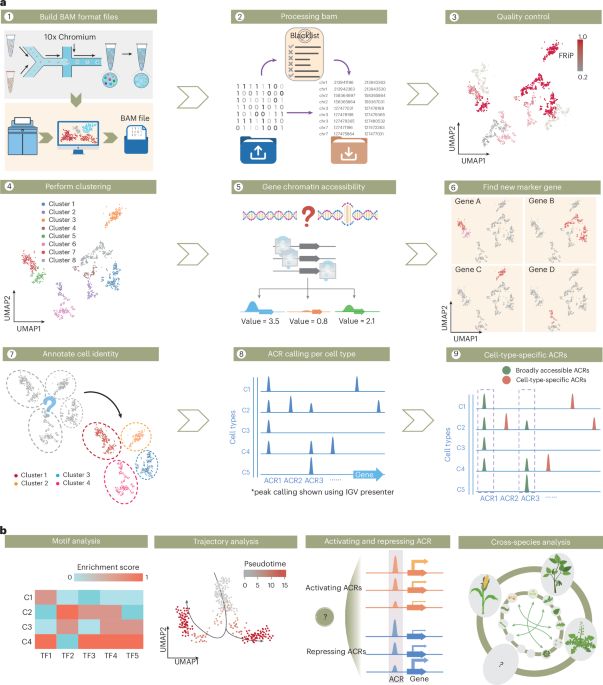
How scSurvival Uses Single Cell Tumor Data to Predict Risk
Researchers supported by the National Institutes of Health recently developed scSurvival, an AI cancer prediction tool built specifically for single cell tumor data. Tested on clinical data from more than 150 cancer patients, the model was able to predict survival outcomes and highlight which cell populations were linked to worse results. In plain terms, it does two things at once: it estimates how likely a patient is to have a poor outcome and points to the particular cells in the tumor that seem responsible. scSurvival works by assigning each tumor cell a weight based on how strongly that cell’s features relate to survival. Cells that appear closely tied to progression or death receive higher weights, while less informative cells are downplayed. The model then combines these weighted signals into a risk score. For patients, being labeled “high-risk” means their tumor contains patterns of cells that, in previous cases, were associated with shorter survival.
What ‘High-Risk’ Really Means for Patients and Doctors
A high-risk label from a tool like scSurvival does not guarantee a bad outcome, but it signals that the tumor’s cell-by-cell makeup matches patterns seen in patients who did worse in the past. That kind of information could reshape conversations between patients and clinicians. Instead of relying only on tumor size, stage, or a few marker tests, doctors could explain which specific cell types are driving concern and why. In practice, high-risk patients might be monitored more closely, offered more intensive therapies, or prioritized for clinical trials evaluating new drugs. Conversely, patients whose tumors look low-risk at the single-cell level may be spared unnecessary side effects from overtreatment. Over time, as AI cancer prediction models are refined and validated in larger groups, they could become an important addition to existing cancer prognosis tools, giving a more personalized and transparent picture of risk.
Beyond Cancer: scPlantReg and the Single-Cell Revolution
The same logic behind scSurvival—reading biology one cell at a time—also applies outside cancer. In plant research, the scPlantReg platform brings together single-cell chromatin accessibility and other data to map cis-regulatory elements, the DNA “switches” that turn genes on and off. By combining methods such as single-cell ATAC-seq with advanced software, scPlantReg helps scientists pinpoint how individual plant cells regulate genes during development, stress, and adaptation. Although plants and tumors are very different, the underlying trend is the same: powerful tools for analyzing single-cell omics data are emerging across biology. Platforms like scATAC-pro, ArchR, and newer unified pipelines are making it easier to handle massive datasets without losing crucial cellular detail. This broader ecosystem of methods and software is important for medicine because many analytical ideas, algorithms, and quality controls developed in one field can be adapted to others, accelerating progress in personalized cancer treatment.
What This Could Mean for Future Diagnosis and Treatment
If single-cell AI models like scSurvival continue to prove reliable, they could change several stages of cancer care. Earlier in the journey, a biopsy could be profiled cell by cell to detect aggressive patterns that are invisible to standard tests, flagging patients who need urgent, intensive treatment. For therapy selection, understanding which high-risk cell populations dominate a tumor could guide targeted drugs or drug combinations aimed at those cells, making personalized cancer treatment far more precise. In clinical trials, enrolling patients based on single-cell risk profiles might help test therapies in the people most likely to benefit, speeding up results and reducing failed studies. However, these tools are still largely in the research phase. They must be validated in larger, more diverse patient groups and integrated with existing workflows before appearing widely in clinics, which is likely to take several years rather than months.